ACNFP Advice on the safety of synthetic cannabidiol (CBD) as a novel food for use in food supplements.
On this page
Skip the menu of subheadings on this page.Reference Number RP07
Regulated Product Dossier Assessment
Assessment finalised: 29th of April 2024
Summary
An application was submitted to the Food Standards Agency (FSA) and Food Standards Scotland (FSS) in January 2021 from Chanelle McCoy CBD Ltd. (“the applicant”) for the authorisation of synthetic cannabidiol (CBD) as a novel food. The novel food is a synthetic >98% pure form of CBD which is intended to be used as a food supplement for adults.
For CBD, a provisional Acceptable Daily Intake (ADI) of 10 mg/day has been published by the FSA, and was considered in assessing this novel food. The provisional ADI (section 2.7) was recommended, subject to the existing advice to consumers that pregnant and breastfeeding women and people taking any prescription medication should avoid the consumption of CBD. Consumers on regular medications should seek advice from a medical professional before using any type of CBD food product. In addition, children and prospective parents trying for a baby are advised against consumption of CBD, as are those who are immunosuppressed, due to remaining data gaps and residual uncertainties concerning the safety of CBD for these groups of consumers. These contraindications would also apply to this novel food.
To support the FSA and FSS in their evaluations of the application, the Advisory Committee on Novel Foods and Processes (ACNFP) were asked to review the safety dossier and supplementary information provided by the applicant. The Committee did not consider any potential health benefits or claims arising from consuming the food, as the focus of the novel food assessment is to ensure the food is safe, and not putting consumers at a nutritional disadvantage.
The Committee concluded that the applicant had provided sufficient information to assure the novel food, a synthetic CBD as outlined in application RP07, was safe under the proposed conditions of use. The anticipated intake levels and the proposed use of this pure form of CBD in foods and food supplements was not considered to be nutritionally disadvantageous.
1. Introduction
1. The ACNFP assessed the food safety risks of synthetic CBD and its production under the proposed uses, in line with Article 7 of assimilated Commission Regulation (EU) 2017/2469. The regulatory framework and the technical guidance put in place by the European Food Safety Agency (EFSA) for full novel food applications is retained as the basis and structure for the assessment (EFSA NDA Panel, 2016).
2. An application was submitted to the Food Standards Agency (FSA) and Food Standards Scotland (FSS) in January 2021 from Chanelle McCoy CBD Ltd. (“the applicant”) for the authorisation of synthetic cannabidiol (CBD) as a novel food. The novel food is a synthetic >98% pure form of CBD which is intended to be used as a food supplement for adults.
3. Advice was sought from the joint Subgroup of the ACNFP and the Committee on Toxicity (COT) on CBD and hemp derived products, on the quality of the toxicological evidence submitted to support the application. The ACNFP and COT have issued a statement on a provisional ADI that can be applied to CBD ingredients containing 98% or more CBD. This, and wider evidence available in the public domain, was taken into account in reviewing the toxicological evidence for this application.
4. The final advice from the Committee was agreed at the 164th meeting, allowing the FSA and FSS to complete the risk assessment.
5. This document outlines the conclusions of the ACNFP on the safety of a synthetic >98% pure form CBD (as detailed in application RP07) as a novel food.
2. Assessment
2.1 Identity of novel food
6. The novel food is a synthetic >98% pure form of CBD which is a white to slightly beige crystalline powder of experimentally derived 100%± 2% purity. CBD is found in natural form in hemp as the (-)- trans isomer. As this CBD was synthesised chemically, chirality analysis of the compound by a commercially validated chiral HPLC method demonstrated a chiral purity of no less than 99.5% (-) - trans cannabidiol. Information to support stereoselective synthesis, and characterisation, was provided for six batches of the novel food.
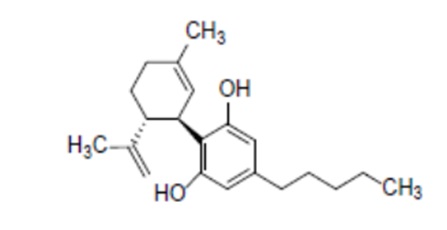
7. CBD in this application is characterised by the chemical formula: C21H30O2; molecular mass: 314.46 g/mol; CAS number: 13956-29-1; isomer: (-)-trans-cannabidiol; IUPAC name: 2-[(1R,6R)-3-methyl-6-prop-1-en-2-ylcyclohex-2-en-1-yl]-5-pentylbenzene-1,3-diol.
Diagram 1: The molecular structure of CBD
8. Confirmation of its identity was provided by Infrared (IR) spectroscopy, 1H Nuclear Magnetic Resonance (NMR) spectroscopy, mass spectrometry, and Ultraviolet visualisation (UV-Vis).
2.2 Production Process
9. The synthetic >98% pure form of CBD is manufactured using a stereoselective multi-step process under controlled conditions.
10. Certificates of analysis for raw starting materials used in the chemical synthesis were provided to demonstrate the effectiveness of the controls at this point in the process.
11. This process begins with olivetol and menthadienol as the primary starting materials, which undergo mixing, filtration, and distillation steps with catalysts present, resulting in the formation of the desired synthetic >98% pure CBD form.
12. For optional purification steps for commercial purposes, the crude final CBD ingredient is washed and separated several times using various processing aids.
13. The ACNFP considered whether the use of solvents as processing aids left any residues that needed to be flagged to risk managers. Comparison was made to residue limits for other consumed products as detailed in Table 1. Residues of solvents have been included in the specification.
Table 1: Comparison of information on permitted residue levels for solvents used in the novel foods production compared to the proposed specification.
|
Solvent used |
Available data on safe maximum level of consumption |
Level in specification for the novel food |
|
Dichloromethane |
Guidance on residues in Pharmaceutical products sets a Permissible Daily Exposure of 6.0 mg/day or at a concentration of 600 ppm1 Permitted extraction solvent residue on coffee at 2mg/kg and 5 mg/kg in tea2 |
600 mg/kg CBD |
|
Methanol |
Guidance on residues in pharmaceutical products sets Permissible Daily Exposure of 30.0 mg/day or a concentration of 3000 ppm.3 Extraction solvent residue maximum level of 10mg/kg product for all food uses2. |
3000 mg/kg CBD |
|
Isopropanol |
Extraction solvent residue maximum level of 10 mg/kg2 ADI of 2.4 mg/kg bw as acceptable previous cargoes for edible fats and oils.4 |
5000 mg/kg CBD |
|
n- heptane |
Used as an extraction solvent, but only in accordance with good manufacturing practices, which should result in minimal residue4 |
5000mg/kg CBD |
1 Q3C (R8) Step 5 - impurities: guideline for residual solvents (europa.eu)
2 Directive 2009/32/EC of the European Parliament and of the Council of 23 April 2009 on the approximation of the laws of the Member States on extraction solvents used in the production of foodstuffs and food ingredients
3 Q3C (R8) Step 5 - impurities: guideline for residual solvents (europa.eu)
4 Scientific Opinion on the evaluation of the substances currently on the list in the annex to Commission Directive 96/3/EC as acceptable previous cargoes for edible fats and oils – Part II of III
14. The evidence presented (see Table 2 below) on composition shows the residues of solvents in the product were consistently below the level set in the specification. When considered at the level of consumption, the evidence suggests the levels of solvent residues in the novel food are below those which would represent a safety concern.
15. A HACCP statement was provided along with further details of the process and how it operates. The production process has characterised the potential hazards and detailed the corresponding control measures sufficiently.
2.3 Compositional Information
16. Results from six independent batches of the novel food demonstrated that the CBD content was being produced consistently (Table 2).
Table 2: Compositional analysis of representative batches of CBD
|
Parameter |
Method of Analysis |
Batch 1 |
Batch 2 |
Batch 3 |
Batch 4 |
Batch 5 |
Batch 6 |
|
Identity and Composition |
|
|
|
|
|
|
|
|
Appearance |
Visual |
White crystalline powder |
White crystalline powder |
Almost white crystalline powder |
White to slightly beige crystalline powder |
White to slightly beige crystalline powder |
White to slightly beige crystalline powder |
|
Identification by IR |
USP 197 |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
|
Identification by HPLC |
In house method |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
Corresponds to reference |
|
Water content (%) |
USP 921 |
≤0.05 |
≤0.05 |
≤0.05 |
0.0 |
0.2 |
0.0 |
|
Residue on ignition (%) |
USP 281 |
≤0.05 |
≤0.05 |
≤0.05 |
0.1 |
0.1 |
0.0 |
|
Specific optical rotation (degrees) |
USP 781 |
-128 |
-130 |
-132 |
-129 |
-129 |
-129 |
|
CBD (%) |
HPLC (in house method) |
99.4 |
99.8 |
99.9 |
100.5 |
100.7 |
100.2 |
|
Olivetol content (%) |
HPLC (in house method) |
≤0.03 |
≤0.03 |
≤0.03 |
≤0.03 |
≤0.03 |
≤0.03 |
|
4-monobromo-cannabidiol (%) |
HPLC (in house method) |
≤0.03 |
≤0.03 |
≤0.03 |
≤0.03 |
≤0.03 |
≤0.03 |
|
Delta-9-tetrahydrocannabidiol (%) |
HPLC (in house method) |
≤0.02 |
≤0.02 |
≤0.02 |
≤0.02 |
≤0.02 |
≤0.02 |
|
Individual unspecified impurities (%) |
HPLC (in house method) |
≤0.05 |
≤0.05 |
≤0.05 |
≤0.05 |
≤0.05 |
≤0.05 |
|
Total impurities (%) |
HPLC (in house method) |
≤0.05 |
≤0.05 |
≤0.05 |
≤0.05 |
≤0.05 |
≤0.05 |
|
Residues (mg/kg of CBD) |
|
|
|
|
|
|
|
|
Methanol |
GC (in house method) |
≤300 |
≤300 |
≤300 |
NM |
NM |
NM |
|
n-heptane |
GC (in house method) |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
|
Dichloromethane |
GC (in house method) |
≤60 |
≤60 |
≤60 |
≤60 |
≤60 |
≤60 |
|
Triethylamine |
GC (in house method) |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
|
Isopropanol |
GC (in house method) |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
|
Isooctane |
GC (in house method) |
≤500 |
≤500 |
≤500 |
≤500 |
≤500 |
545 |
GC = Gas chromatography; HPLC = high-performance liquid chromatography; IR = infrared spectroscopy; NM = not measured; USP = United States Pharmacopeia.
17. CBD content is consistently above 98% pure with negligible amounts of starting materials detected across the six representative batches.
18. It is recognised that the detection and characterisation of cannabinoids in a range of food matrices is an evolving area and there are yet to be internationally recognised methods. The limitations of analytical methodology available have been subject to discussion in the Joint ACNFP and COT CBD Subgroup and remain a source of uncertainty in the assessment. As a result, the robustness, accuracy and precision of the methods have been considered in interpreting the data on THC and were considered appropriate in this case.
19. Delta-9-tetrahydrocannabidiol (THC), as a potential contaminant in the novel food, was detected at levels at or below 0.02 mg/kg CBD in all six batches tested.
20. A literature review was undertaken as part of the assessment of CBD as a novel food, to understand the impact on the safety of foods with trace levels of contamination with THC. The joint ACNFP and COT subgroup reviewed the information from literature and identified a point of departure from the European Food Safety Authority (EFSA) opinion on THC as a contaminant in milk and meat (EFSA,2015). The point of departure was a LOAEL (Lowest Observed Effect Level) of 0.036mg/kg/bw/day from the most sensitive individuals and the lowest dose tested in the human clinical studies reviewed.
21. Uncertainty factors, including a factor of 3 to extrapolate from LOAEL to a NOAEL (No Observed Adverse Effect Level) were applied to the LOAEL identified. This was considered appropriate as the effects are mild to moderate in severity. A further factor of 10 was applied for person to person variation resulting in a total uncertainty factor of 30. The factor of 30 applied to the 0.036mg/kg/bw/day LOAEL results in a level of 1µg /kg bw/day that would be represent a safe upper intake for consumed THC as a contaminant in food. This was identified an acute reference dose (ARfD) (EFSA, 2015)
22. The Subgroup agreed the ARfD to be sufficiently protective to apply to the UK population. It was noted that in applying the acute reference dose EFSA have assumed that the effects seen would be the same if humans were exposed to multiple doses of THC at very low levels. The Subgroup commented that there was not data to verify this assumption, but if setting limits the dataset is the best available.
23. The levels of THC in the novel food once adjusted to take into account of the proposed uses - 10mg of CBD being consumed a day, were below the ARfD identified by EFSA of 1µg /kg bw/day or 70µg/day for a healthy adult. This level does not present a concern in terms of consumer safety for the novel food under the proposed conditions of use.
24. To ensure THC levels remain consistently low in the production of CBD, THC should be a standard substance included in the specification as relevant to all batches produced.
25. Analytical data, presented as the mean of three independent batches of the novel food, demonstrated that heavy metals where detected were present in very low levels and were below established UK regulatory limits where applicable (arsenic, cadmium, mercury and lead) (Table 3).
Table 3: The content of heavy metals, minerals and trace elements in the novel food
|
Parameter (mg/kg) |
Maximum concentration measured (mg/kg) |
|
Arsenic |
≤0.1 |
|
Cadmium |
≤0.1 |
|
Mercury |
≤0.1 |
|
Lead |
≤0.1 |
|
Cobalt |
≤0.1 |
|
Nickel |
0.2 |
|
Vanadium |
≤0.1 |
|
Silver |
≤0.1 |
|
Gold |
≤0.2 |
|
Iridium |
≤0.1 |
|
Osmium |
≤0.7 |
|
Palladium |
≤0.1 |
|
Platinum |
≤0.1 |
|
Rhodium |
≤0.1 |
|
Ruthenium |
≤0.1 |
|
Selenium |
≤0.1 |
|
Thallium |
≤0.1 |
|
Barium |
≤1 |
|
Chromium (3) |
0.1 |
|
Copper |
0.1 |
|
Lithium |
≤0.1 |
|
Molybdenum |
≤0.4 |
|
Antimony |
≤0.1 |
|
Tin |
≤0.1 |
26. Analytical data concerning the microbiological content from three independent batches of the novel food were reported (Table 4). The process in manufacturing this novel food uses extreme high and low temperatures, harsh pH conditions and alcohol solvents. Full microbial risk assessment as per USP 61 and USP 62 confirm that the novel food does not raise a safety concern and consistently meets the proposed microbial specification levels.
Table 4: The microbiological analysis of the novel food
|
Parameter |
Batch 1 |
Batch 2 |
Batch 3 |
|
Total aerobic microbial plate count |
< 100 CFU/g |
< 100 CFU/g |
< 100 CFU/g |
|
Total yeast and mould plate count |
< 100 CFU/g |
< 100 CFU/g |
< 100 CFU/g |
27. Additionally, water activity as measured in aw values, of three representative samples of the novel food was measured as per USP 1112. The results further demonstrate that microbial growth in the CBD novel food is unlikely at the low aw values (below 0.6) for the final ingredient (Table 5). Therefore, microbial analysis is not a required parameter for inclusion in the specification.
Table 5: Water activity analysis
|
Batch |
Result aw |
|
1 |
0.40 |
|
2 |
0.40 |
|
3 |
0.27 |
|
Average |
0.36 |
28. The data presented indicated the novel food was appropriately characterised.
2.4 Stability
29. The stability of the synthetic CBD was assessed under real-time conditions (25oC and 60% relative humidity) and under refrigerated conditions (5oC) in 3 batches for 3 months, in a further 3 batches for 6 months and another 3 batches for 9 months. Results showed that the novel food meets the specification criteria for CBD content, and no changes in appearance, water content and impurity levels are seen over these time periods. The THC content was also tested using HPLC and remained consistently below 0.05% w/w across the time period which is below the 0.10%w/w in the specification.
30. The stability of the synthetic CBD was assessed under accelerated conditions (40oC and 75% relative humidity) in six batches over a maximum period of 9 months. Results confirmed that the novel food meets the specification criteria for CBD content and no changes in appearance, water content and impurity levels are seen over these time periods over this time period.
31. The data provided supports the stability of the novel food for a period of at least 9 months.
2.5 Specification
32. The specification parameters reported in Table 6 were assessed using internationally recognised methods or determined using internally developed and validated methods. The data in Table 2 demonstrates the novel food can be produced consistently to the proposed specification.
Table 6: Specification of the novel food
|
Description |
|
Cannabidiol is a white to slightly beige powder produced by chemical synthesis |
|
Parameter |
Specification |
Method |
|
Appearance |
White to slightly beige crystalline powder |
Visual inspection |
|
Identity |
Complies |
IR: USP 197 HPLC: in-house method |
|
Water content (%) |
≤0.5 |
USP 921 |
|
Residue on ignition (%) |
≤0.2 |
USP 281 |
|
Specific optical rotation (degrees) |
-140 to -122 |
USP 781 |
|
CBD (%) |
97 to 102 |
HPLC: in house method |
|
Olivetol content (%) |
≤0.15 |
HPLC: in house method |
|
4-monobromo-cannabidiol (%) |
≤0.15 |
HPLC: in house method |
|
Delta-9-tetrahydrocannabidiol (THC) (%) |
≤0.10 |
HPLC: in house method |
|
Individual unspecified impurities (%) |
≤0.15 |
HPLC: in house method |
|
Total impurities (%) |
≤1.0 |
HPLC: in house method |
|
Residual solvents (mg/kg CBD) |
|
|
|
Methanol |
≤3000 |
GC: in house method |
|
n-Heptane |
≤5000 |
GC: in house method |
|
Dichloromethane |
≤600 |
GC: in house method |
|
Triethylamine |
≤5000 |
GC: in house method |
|
Isopropanol |
≤5000 |
GC: in house method |
|
Isooctane |
≤5000 |
GC: in house method |
CBD = cannabidiol; GC = gas chromatography; HPLC = high-performance liquid chromatography; IR = infrared spectroscopy; USP = United States Pharmacopeia
33. Following discussion with the applicant it was agreed that information on microbiological and heavy metal limits did not need to be included in the specification to appropriately characterise the novel food. No specific recommendations were made to amend the specification. The ACNFP concluded the information provided is sufficient to meet the specification of CBD and appropriately characterises the novel food seeking authorisation.
2.6 History of Use
34. Hemp has been widely consumed in the UK and EU as a seed oil, in tea and as an alternative to hops in beer. Extracts of hemp including CBD and synthetic CBD have not been widely consumed and are considered novel foods. While CBD products are widely available on the UK high street, indicating some consumption of CBD as a food, no applications for CBD have yet received authorisation as a novel food.
35. As detailed in the COT review of the literature there has been use of both hemp derived and synthetic forms of CBD for medicinal purposes. These provide an indication of the toxicological effects that should be explored in the testing regime – primarily effects on liver, thyroid and potential impacts on reproductive organs. Also reported, are behavioural effects such as somnolence (sleepiness).
36. As reported in the COT review of the publicly available data on CBD and summary data on a medicinal product, signs of adverse effects on the liver were observed at doses of CBD as low as 5 mg/kg bw/day in patients and healthy human volunteers; this dose is equivalent to 350 mg in a 70 kg adult. The data in the literature also suggested that humans might be more sensitive to the adverse effects of CBD in the liver than laboratory animals.
37. Somnolence effects were noted at doses ≤10 mg/kg bw/day in human studies. Inhibitory drug-drug interactions have also been observed with some medications when CBD is co-administered at doses of 1 mg/kg bw/day (equivalent to 70 mg in a 70 kg adult); the likelihood of effects at lower doses in humans has not been determined.
38. It is noted that the doses of CBD used for medicinal purposes are higher than those proposed for food use. The purpose of an assessment for medicines authorisation is different to that for food and this is reflected in the data requirements. Unlike medicines, there is no risk-benefit context in foods with the requirement instead being that the products are safe. This means that outcomes that are considered to be an adverse event for food might be weighed differently in the context of the clinical benefits in a medicinal study.
39. Within the literature, further human studies utilising chemically derived CBD provides further evidence of a history of synthetic CBD use (Izegelov et al., 2010; Stereo Biotechs Ltd., 2020; Klotz et al., 2019; Wheless et al., 2019). A review by Heuestis et al., 2019 of Cannabidiol Adverse effects and Toxicity notes that, while CBD is not risk-free, severe adverse events occur at doses higher than those recommended for human pharmacotherapies which are prescribed to treat forms of epilepsy.
40. The data on previous consumption of CBD suggest areas for careful consideration in the toxicological review to understand potential effects at the lower doses used in foods.
2.7 Proposed Use and Anticipated Intake
41. The proposed use for the novel food is as a food supplement in the form of an oil sold as capsules and drops at the dose of 10mg per day of CBD. The intended use is food supplements as defined by UK legal requirements (The Food Supplements (England) Regulations 2003) as capsules, liquid or drops in dose form. A provisional acceptable daily intake (ADI) for the use of >98% pure form CBD has been established at 10 mg per day (October 2023) and discussed in the Toxicological information section.
|
Food Category |
Maximum intake level of CBD from product use |
|
Food Supplements (for adults) as defined in the Food Supplements (England) Regulations 2003 as capsules, liquids or drops intended for those 18 years of age or over. |
10mg per day |
42. It is noted that there are already many products available containing CBD. As such, the assessment has been made on the basis of identification of a maximum level of CBD that can be consumed per day per product. As such proposed uses will only be considered safe for all consumers within the assessment when below an intake maximum of 10mg of CBD per day.
43. Risk managers must consider whether consumers would benefit from information on the CBD content of foods in order to ensure their consumption does not exceed a provisional acceptable daily intake of 10mg per day for a healthy adult.
44. As recommended in the ACNFP and COT statement on CBD of >98% purity, “The provisional ADI is recommended, subject to the existing advice to consumers that pregnant and breastfeeding women and people taking any prescription medication should avoid the consumption of CBD. Consumers on regular medications should seek advice from a medical professional before using any type of CBD food product. In addition, children and prospective parents trying for a baby are advised against consumption of CBD, as are those who are immunosuppressed, due to remaining data gaps and residual uncertainties concerning the safety of CBD for these groups of consumers.”
45. The food supplement products are to be labelled in accordance with the labelling requirements of Food Supplements (England) Regulations 2003as follows: Does not exceed the safe limit of 10 mg/day for a 70kg healthy adult. The novel food authorisation should also include the following: Not suitable for use under the age of 18. Not suitable for use during pregnancy or breastfeeding. If you are taking medication or have existing health conditions, please consult your doctor before using this product.
46. The ACNFP explored the potential for foreseeable misuse of the novel food. It is highlighted to risk managers that they may wish to consider whether risk management measures are needed beyond those in the food supplements regulation to ensure consumers are aware of the provisional ADI of 10 mg CBD/day for the product, a dose at which it is considered that no adverse effects would be expected.
2.8 Absorption, Distribution, Metabolism and Excretion (ADME)
47. The Absorption, Distribution, Metabolism and Excretion (ADME) of CBD are known to be complicated by the food matrix in which the CBD is delivered, and are currently still being defined by professional bodies.
48. The oral bioavailability of CBD is low, indicating that it is not absorbed to any notable extent following ingestion (Mechoulam et al., 2002). Published works report the bioavailability of CBD to be between 13 and 19% (Grotenhermen (2003)) or 6% (Hawksworth and McArdle (2004)). The low systemic availability was demonstrated by Martin-Santos et al., 2012 and further supported by a literature search which identified the pharmacokinetics of CBD in humans and concluded with dose dependent peak plasma concentrations of CBD and area under the curve results indicating minimal accumulation (Miller et al., 2018).
49. Following oral consumption, CBD is extensively metabolised in the liver. This rapid first pass metabolism contributes to the low oral bioavailability reported in the literature (Taylor et al., 2018; WHO, 2018). In vitro studies indicate that CYP3A4 and CYP2C19 are the primary hepatic enzymes responsible for first-pass metabolism of CBD; however, several other hepatic cytochrome P450 isoforms (CYP1A1, CYP1A2, CYP2C9, CYP2D6, and CYP3A5) have also demonstrated a capability of metabolising CBD (Jiang et al., 2011; Zendulka et al., 2016).
50. The metabolism of CBD is thought to follow two separate pathways. One is P450-mediated, in which CBD is metabolised into its major metabolite 7-COOH-CBD. This is followed by further metabolic reactions which yield the minor metabolites of CBD, including 6-OH-CBD (Devinsky et al., 2018; Taylor et al., 2018;). The other involves decarboxylation (Kraemer et al., 2019). The resultant metabolites are predominantly excreted in faeces and urine (Hawksworth and McArdle, 2004; WHO, 2018).
51. Accumulation of CBD in plasma of up to 2-fold has been reported when steady state levels are compared with a single dose (such as in Taylor et al., 2018). Additionally, minimal evidence of plasma accumulation has been reported in dosing studies over 5–9 days (Millar et al., 2018; Sellers et al., 2013; Stott et al., 2013).
52. The pharmacokinetics of CBD (24 studies) have also been systematically reviewed by Millar et al., 2018., most of which assessed the administration of CBD at doses of 5–20 mg/day. This correlates to a low dose application similar to this synthetic CBD novel food application. With oral administration, single doses of 5.4 and 10 mg CBD achieved peak serum concentrations (Cmax) of 0.9 and 2.5 ng/ml. The time to maximum concentration (Tmax) was approximately 1 h, with a half-life between 1 and 3 hours. Given the intended use of this synthetic CBD, with an approximate half-life of one to three hours, with a total clearance of 6 hours, there are no significant concerns of accumulation.
53. Based on the information on ADME it is noted that, the bioavailability of CBD when consumed orally is typically low, but can be affected by food matrix. It was noted that the potential for CBD to accumulate in the body has not been examined based on the data supplied. This provides context for interpreting the toxicological data. This also suggests the food context for the novel ingredient could impact whether the CBD present in the ingredient is more or less bioavailable. This has been taken into account in considering the assessment factors to account for uncertainty in setting the provisional ADI.
2.9 Nutritional information
54. The ACNFP sought clarification of the potential for the presence of antinutritional factors from the preparation. As the product is chemically derived from a well-defined synthetic process and characterisation indicated high CBD purity, no antinutritional factors were expected to be present. Information on the composition has confirmed that there is no presence of other components that would impact the digestion or absorption of nutrients from the diet.
55. The data on nutritional composition confirms that CBD has no caloric or nutritional value. The application is not intending that CBD replace another food in the diet. Consumption of the novel food at the proposed use levels is not expected to be nutritionally disadvantageous for consumers.
2.10 Toxicological information
56. Toxicological studies on CBD were performed by the applicant to support the safety assessment of the novel food. The respective study reports are unpublished and claimed as confidential and proprietary data. They were considered essential in the assessment of the safety of the novel food and were reviewed by the ACNFP. How data on systemic toxicity was managed and interpreted in the context of the provisional ADI is explained in the sub-chronic toxicology section below.
2.10.1 Genotoxicity
57. In vitro genotoxicity testing of CBD was conducted under Good Laboratory Practice (GLP) conditions and utilised the following OECD guidelines: in vitro bacterial reverse mutation test (OECD TG 471) and in vitro mammalian cell micronucleus test (OECD TG 487). This approach is recommended by the UK Committee on Mutagenicity and is also the basis of guidance on the preparation and submission of an application for authorisation of a novel food in the context of Regulation (EU) 2015/2283.
58. The in vitro bacterial reverse mutation test [(Gijsbrechts, 2020 (unpublished)] demonstrated that this CBD ingredient was non-mutagenic in the absence and presence of metabolic activation. In addition, the in vitro mammalian cell micronucleus test [(de Jong, 2020 (unpublished)] demonstrated that cannabidiol was non-clastogenic and non-aneugenic in the absence and presence of metabolic activation.
59. The results from these in vitro studies support the conclusion that the novel food (>98% pure CBD) is not genotoxic. This is consistent with the view of the Committee on Mutagenicity in reviewing CBD generically from evidence available in the public domain (Committee on Mutagenicity; MUT/MIN/2020/1, 2020).
2.10.2 Sub-chronic toxicological study
60. The joint subgroup of the ACNFP and COT was formed to address a series of questions in relation to the safety of CBD, cannabinoids and hemp-derived ingredients.
61. A weight of evidence approach has allowed the Subgroup to identify a provisional ADI for CBD ingredients, of >98% purity, of 0.15mg/kg bw/day or 10 mg per day for a 70 kg healthy adult (Joint position paper from the ACNFP and COT; FSA consumer advice published in October 2023 ). This value was identified to be protective of the most sensitive known effects in the liver and thyroid parameters, and included consideration of data gaps and uncertainties.
2.10.3 Sub-chronic data supplied for this application
62. This applicant provided a Repeated Dose 90-Day Oral Toxicity Study in Rodents ([McGeoghie, 2020 unpublished)], which was conducted under GLP conditions and to OECD Technical Guideline 408. In this 90-day study, each group comprised 10 female and 10 male rats which were dosed with 0 (control – corn oil), 15, 36 or 72 mg/kg bw/day CBD once per day by oral gavage at a dose volume of 4mL/ kg / body weight .
63. Review of the study supported the conclusion that it was of sufficient quality to support the safety of the novel food. The findings of the study were consistent with those considered in the development of the provisional ADI . It was, therefore, considered scientifically appropriate to apply the provisional ADI of 0.15mg/kg bw/day or 10mg/day as identified in the joint statement of the ACNFP and COT on >98% pure forms of CBD.
2.11 Allergenicity
64. This synthetic CBD comprises >98% pure CBD and the production process for CBD does not introduce any risk of allergenic potential. CBD as a chemical entity, suggests the potential for IgE mediated food allergy is unlikely.
65. The assessment considered whether the remaining 2% of the novel food composition was likely to be allergenic or elicit food allergic reactions. It was noted that none of the raw materials or processing aids used in the production process are derived from or contain any of the allergenic food ingredients specified under Regulation (EU) No 1169/2011 of the European Parliament and of the Council of 25 October 2011 on the provision of food information to consumers. Suggesting the potential to elicit reactions in those sensitive to those foods is unlikely.
66. The novel food is unlikely to trigger allergic reactions in the target population under the proposed conditions of use.
3. Discussion
67. The novel food is a synthetic CBD containing 100% ± 2% CBD (a Group A CBD of >98% purity), chemically produced using a multi-step manufacturing process.
68. This synthetic CBD is intended to be used a food supplement for adults as an oil or as an oil in capsules at a dose of 10mg a day; it is not intended to replace any food.
69. In October 2023, the Joint ACNFP and COT subgroup identified a provisional acceptable daily intake (ADI) of 10 mg per day (0.15 mg/kg bw/day) for CBD novel foods containing 98% CBD or above, such as the novel food discussed in this assessment.
70. A weight of evidence approach was used to arrive at a provisional ADI of 10 mg/day (0.15 mg/kg bw/day). The most sensitive human health effects, that this ADI protects against, are seen consistently in the liver and thyroid in a number of studies using >98% pure CBD. This value also takes account of the lack of human-based long-term evidence and evidence regarding potentially vulnerable groups, which is applied here for this CBD isolate(footnote).
71. Based upon the dossier of evidence provided by the applicant, the safety of the novel food was reviewed and evidence to reach a conclusion on safety provided. The evidence presented is consistent with evidence presented to support the development of a provision ADI of 10 mg/day for CBD of 98% purity or above. As such the provisional ADI should be applied to this novel food.
72. This is subject to the existing advice to consumers that pregnant and breastfeeding women and people taking any prescription medication should avoid the consumption of CBD. Consumers on regular medications should seek advice from a medical professional before using any type of CBD food product. In addition, children and prospective parents trying for a baby are advised against consumption of CBD, as are those who are immunosuppressed, due to remaining data gaps and residual uncertainties concerning the safety of CBD for these groups of consumers. These contraindications would also apply to this novel food.
73. The maximum safe exposure for healthy adults of 70kg as identified in the provisional ADI is 10mg per day. If the inclusion level of this CBD isolate leads to an intake per individual serving of each product type of 10 mg/day, only one product type per day should be consumed to ensure the provisional ADI is not exceeded. Multiple intakes of products containing CBD on the same day should be avoided to support minimising exposure to below the provisional ADI.
4. Conclusions
74. The ACNFP have undertaken a review of this synthetic CBD and concluded that the novel food is safe under the proposed conditions of use and does not pose a safety risk to human health. The proposed uses are not considered nutritionally disadvantageous.
75. These conclusions were based on the information in the novel food dossier submitted by the applicant plus the supplementary information from a wider review of CBD and cannabinoids and could not have been reached without the following data claimed as proprietary by the applicant:
- annexes to the dossier which relate to the identity of the novel food, the production process, stability, methods of analysis, particle size analysis and toxicology.
- in vitro bacterial reverse mutation test [(Gijsbrechts, 2020 (unpublished)], in vitro mammalian cell micronucleus test [(de Jong, 2020 (unpublished)] and 90-day repeat dose feeding study ([McGeoghie, 2020 (unpublished)].
76. The members of the ACNFP during the course of the assessment who were;
Dr Camilla Alexander White, Dr Anton Alldrick, Dr Kimon Andreas Karatzas, Alison Austin, Professor George Bassel, Dr Mark Berry, Dr Christine Bosch, Professor Dimitris Charalampopoulos, Dr Catharina Edwards, Professor Susan Fairweather-Tait, Professor Paul Frazer, Dr Hamid Ghoddusi, Professor Andy Greenfield, Professor Wendy Harwood, Professor Huw D. Jones, Dr Ray Kemp, Dr Elizabeth Lund, Professor Harry J. McArdle, Mrs Rebecca McKenzie, Professor Clare Mills, Dr Antonio Peña-Fernández, Dr Lesley Stanley, Professor Hans Verhagen,
Dr Maureen Wakefield, and Professor Bruce Whitelaw.
77. To note, interests were received from members of the ACNFP, Dr Alldrick declared a potential interest relating to his previous employment and this was considered a potential conflict and as a result he was not present for discussions of CBD by the Committee. Emeritus Prof Harry McArdle declared an interest from his work with EFSA’s novel food Committee in considering data requirements for CBD. While not seen as a conflict, to avoid Prof McArdle being subject to information that would influence his EFSA work, it was agreed that he would not be present in discussions for CBD by the Committee but could supply comments for consideration by the Committee upon review of the minutes.
5. References
Advisory Committee on Novel Foods and Processes (ACNFP) and Committee on Toxicity (COT), 2023. Joint position paper from the Advisory Committee on Novel Foods and Processes (ACNFP) & Committee on Toxicity (COT) on establishing a provisional acceptable daily intake (ADI) for pure form (≥98%) cannabidiol (CBD) in foods, based on new evidence. Published October 2023 DOI: https://doi.org/10.46756/sci.fsa.zcg392
Committee On Mutagenicity Of Chemicals In Food, Consumer Products And The Environment, (MUT/MIN/2020/1), 2020; Committee on Mutagenicity Metting 2020/1
de Jong BG, 2020 [unpublished]. Prepared by Charles River Laboratories Den Bosch B.V., DD ‘s Hertogenbosch, The Netherlands for Chanelle McCoy CBD Ltd., Loughrea, Galway, Ireland. Study Title: Cannabidiol: in vitro mammalian cell micronucleus test. Confidential. (Test Facility Study No. 20203946).
Devinsky O, Patel AD, Thiele EA, Wong MH, Appleton R, Harden CL, Greenwood S, Morrison G and Sommerville K, 2018. Randomized, dose-ranging safety trial of cannabidiol in Dravet syndrome [GWPCARE1 Part A Study Group]. Neurology, 90, e1204-e1211.
EFSA CONTAM Panel (Panel on Contaminants in the Food Chain), 2015. Scientific opinion on the risks for human health related to the presence of Tetrahydrocannabidiol (THC) in milk and other food from animal origin. DOI , 10.2903/j.efsa.2015.4141
EFSA NDA Panel (EFSA Panel on Nutrition and Novel Foods). (2016). Guidance on the Preparation and Presentation of an Application for Authorisation of a Novel Food in the Context of Regulation (EU) 2015/2283. EFSA Journal, 14(11), 4594. Https://doi.org/10.2903/j.efsa.2016.4594
Gijsbrechts JJA, 2020 [unpublished]. Prepared by Charles River Laboratories Den Bosch B.V., DD ‘s Hertogenbosch, The Netherlands for Chanelle McCoy CBD Ltd., Loughrea, Galway, Ireland. Study Title: Cannabidiol: bacterial reverse mutation test. Confidential. (Test Facility Study No. 20203944).
Grotenhermen, F. (2003) ‘Pharmacokinetics and pharmacodynamics of cannabinoids’, Clinical Pharmacokinetics, 42(4), 327–360. https://doi.org/10.2165/00003088-200342040-00003
Hawksworth, G. and McArdle, K. (2004) ‘Metabolism and pharmacokinetics of cannabinoids’, in Guy, G, Whittle, B and Robson, P (eds.), The Medicinal Uses of Cannabis and Cannabinoids. London: London Pharmaceutical Press. pp. 205–228. Google Scholar
Huestis MA, Solimini R, Pichini S, Pacifici R, Carlier J, Busardò FP. Cannabidiol Adverse Effects and Toxicity. Curr Neuropharmacol. 2019;17(10):974-989. doi: 10.2174/1570159X17666190603171901
Jiang R, Yamaori S, Takeda S, Yamamoto I and Watanabe K, 2011. ‘Identification of cytochrome P450 enzymes responsible for metabolism of cannabidiol by human liver microsomes’, Life Sciences, 89(5–6), 165–170. doi:10.1016/j.lfs.2011.05.018.
Joint position paper from the Advisory Committee on Novel Foods and Processes (ANCFP) & Committee on Toxicity (COT) on establishing a provisional acceptable daily intake (ADI) for pure form (≥98%) cannabidiol (CBD) in foods, based on new evidence; October 2023. https://doi.org/10.46756/sci.fsa.zcg392
Izgelov D, Davidson E, Barasch D, Regev A, Domb AJ, Hoffman A. Pharmacokinetic investigation of synthetic cannabidiol oral formulations in healthy volunteers. Eur J Pharm Biopharm. 2020 Sep;154:108-115. doi: 10.1016/j.ejpb.2020.06.021
Klotz KA, Grob D, Hirsch M, Metternich B, Schulze-Bonhage A, Jacobs J. Efficacy and Tolerance of Synthetic Cannabidiol for Treatment of Drug Resistant Epilepsy. Front Neurol. 2019 Dec 10;10:1313. doi: 10.3389/fneur.2019.01313
Martin-Santos, R., Crippa, J. A., Batalla, A., Bhattacharyya, S., Atakan, Z., Borgwardt, S., Allen, P., Seal, M., Langohr, K., Farré, M., Zuardi, A. W and McGuire, P. K. (2012). Acute effects of a single, oral dose of d9-tetrahydrocannabinol (THC) and cannabidiol (CBD) administration in healthy volunteers. Current pharmaceutical design, 18(32), 4966–4979. https://doi.org/10.2174/138161212802884780
‘MDA’, Misuse of Drugs Act 1971, UK Public General Acts1971 c. 38, Misuse of Drugs Act 1971 (legislation.gov.uk)
Mechoulam R, Parker LA and Gallily R, 2002. Cannabidiol: an overview of some pharmacological aspects. Journal of Clinical Pharmacology, 42, 11S-19S.
Millar SA, Stone NL, Yates AS and O'Sullivan SE, 2018. A Systematic Review on the Pharmacokinetics of Cannabidiol in Humans. Frontiers in Pharmacology, 9, 1365 [13pp, plus supplementary table]. doi: 10.3389/fphar.2018.01365
OECD (Organisation for Economic Co-operation and Development), 1997. Bacterial reverse mutation test. In OECD guidelines for the testing of chemicals. OECD guideline No 471 (updated & adopted: 21 July 1997). Paris, France: Organisation for Economic Co-operation and Development (OECD).
https://doi.org/10.1787/9789264071247-en
OECD (Organisation for Economic Co-operation and Development), 1998. OECD principles of good laboratory practice. Series on principles of good laboratory practice and compliance monitoring, No. 1 (ENV/MC/CHEM(98) 17). Paris, France: Organisation for Economic Co-operation and Development (OECD), Environment
Directorate, Chemicals Group and Management Committee.
https://doi.org/10.1787/9789264078536-en
OECD (Organisation for Economic Co-operation and Development), 2016. In vitro mammalian cell micronucleus test. In OECD guidelines for the testing of chemicals. OECD guideline No 487 (updated & adopted: 29 July 2016). Paris, France: Organisation for Economic Cooperation and Development (OECD).
https://doi.org/10.1787/9789264264861-en
OECD (Organisation for Economic Co-operation and Development), 2018. Repeated dose 90-day oral toxicity study in rodents. In OECD guidelines for the testing of chemicals. OECD guideline No 408 (updated and adopted 27 June 2018). Paris, France: Organisation for Economic Cooperation and Development (OECD).
https://doi.org/10.1787/9789264070707-en
McGeoghie S, 2020 [unpublished]. Prepared by Charles River Laboratories, East Lothian, UK for Chanelle McCoy CBD Ltd., Loughrea, Galway, Ireland. Study Title: Cannabidiol: a 90-day toxicity study by oral (gavage) in the rat. Confidential. (Test Facility Study No. 509549)
Sellers EM, Schoedel K, Bartlett C, Romach M, Russo EB, Stott CG, Wright S, White L, Duncombe P, Chen CF. A Multiple-Dose, Randomized, Double-Blind, Placebo-Controlled, Parallel-Group QT/QTc Study to Evaluate the Electrophysiologic Effects of THC/CBD Spray. Clin Pharmacol Drug Dev. 2013 Jul;2(3):285-94. doi: 10.1002/cpdd.36
Stott CG, White L, Wright S, Wilbraham D, Guy GW. A phase I study to assess the single and multiple dose pharmacokinetics of THC/CBD oromucosal spray. Eur J Clin Pharmacol. 2013 May;69(5):1135- 47. doi: 10.1007/s00228-012-1441-0
Stero Biotechs Ltd. Phase 2a Study to Evaluate the Safety, Tolerability and Efficacy of Cannabidiol as a Steroid-sparing Therapy in Steroid-dependent Crohn's Disease Patients. ClinicalTrials.gov Identifier: NCT04056442. Last Update Posted : June 30, 2020. Available online at: A Phase 2a Study to Evaluate the Safety, Tolerability and Efficacy of Cannabidiol as a Steroid-sparing Therapy in Steroid-dependent Crohn's Disease Patients - Full Text View - ClinicalTrials.gov
The Food Supplements (England) Regulations 2003, No. 1387, England
The Food Supplements (Wales) Regulations 2003 (legislation.gov.uk)
The Food Information Regulations 2014, No.1855
Taylor L, Gidal B, Blakey G, Tayo B, Morrison G. A Phase I, Randomized, Double-Blind, Placebo Controlled, Single Ascending Dose, Multiple Dose, and Food Effect Trial of the Safety, Tolerability and Pharmacokinetics of Highly Purified Cannabidiol in Healthy Subjects. CNS Drugs. 2018 Nov;32(11):1053-1067. doi: 10.1007/s40263-018-0578-5
Wheless JW, Dlugos D, Miller I, Oh DA, Parikh N, Phillips S, Renfroe JB, Roberts CM, Saeed I, Sparagana SP, Yu J, Cilio MR; INS011-14-029 Study Investigators. Pharmacokinetics and Tolerability of Multiple Doses of Pharmaceutical-Grade Synthetic Cannabidiol in Pediatric Patients with Treatment-Resistant Epilepsy. CNS Drugs. 2019 Jun;33(6):593-604. doi: 10.1007/s40263-019-00624-4
Zendulka, O., Dovrtělová, G., Nosková, K., Turjap, M., Šulcová, A., Hanuš, L and Juřica, J. (2016). Cannabinoids and Cytochrome P450 Interactions. Current drug metabolism, 17(3), 206–226. https://doi.org/10.2174/1389200217666151210142051
Abbreviations
| 1H NMR | 1H (proton) nuclear magnetic resonance |
| ACNFP | Advisory Committee on Novel Foods and Processes |
| ADI | Acceptable Daily Intake |
| ADME | Absorption, Distribution, Metabolism and Excretion |
| ARfD | Acute Reference Dose |
| aw | Water activity |
| bw | body weight |
| CAS | Chemical Abstracts Service |
| CBD | Cannabidiol |
| Cmax | Peak serum concentration |
| COT | Committee on Toxicity |
| CFU | Colony Forming Unit |
| EC | European Commission |
| EFSA | European Food Safety Agency |
| EMA | Environmental Medicines Agency |
| EU | European Union |
| FDA | Food and Drug Administration (USA) |
| FSA | Food Standards Agency |
| FSS | Food Standards Scotland |
| GC | Gas chromatography |
| GLP | Good Laboratory Practice |
| HACCP | Hazards Analysis and Critical Control Points |
| HPLC | High-performance liquid chromatography |
| IR | Infra-red |
| LOAEL | Lowest Observable Adverse Effect Level |
| NOAEL | No Observable Adverse Effect Level |
| NM | Not measured |
| OECD | Organisation for Economic Co-operation and Development |
| Tmax | Time to maximum concentration |
| USP | United States Pharmacopeia |
| UV | ultra-violet |